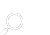USA Professor Discovers Natural Ionic Liquid; Finding Could Impact Development and Use of Synthetic Form
Posted on October 1, 2014
A University of South Alabama professor has discovered a naturally occurring ionic liquid, a category of liquid salt that has been synthetically engineered for industrial purposes and now is known to come from another source – fighting ants.
The discovery by Dr. James Davis, professor of chemistry, could impact the development and use of synthetic ionic liquids, which are used as solvents for chemical reactions, as electrolytes in batteries, as high-tech lubricants, as propellants for satellite maneuvering, and increasingly people are looking to formulate medicines into ionic liquid forms to aid in drug dosing and delivery.
Davis began his research after reading an article in Chemical & Engineering News on work with rival ant populations conducted by researchers at the University of Texas. Tawny crazy ants have been displacing fire ants in certain areas, despite the former’s smaller size and weaker venom.
When sprayed by a fire ant’s poison, according to the article, the tawny crazy ant retreats and bathes itself in its own venom – formic acid – before heading back into battle. The formic acid, the UT researchers found, somehow protects the tawny crazy ant and greatly increases its chances of survival.
Davis is a leading chemist in the field of ionic liquids. Based on his background, Davis said, one look at the molecular structure of the fire ant venom illustrated in the article was all it took to raise his suspicion about what was happening – that the alkaloid-based fire ant venom was mixing with the formic acid to create an ionic liquid.
To test his hypothesis, Davis asked fellow scientists from Auburn University and in China to send him samples of fire ant venom. Davis and his students then took to the lab and mixed the venom with formic acid, resulting in a viscous ionic liquid.
“If ionic liquids exist in nature, they exist for purpose. They have some biological function,” Davis said. “If we can study that, if we can find new ones, if we can figure out what those biological roles are, we can then take that information, harness it and use it potentially to develop new forms of drugs and medicines that might be more effective.”
Davis said he is not surprised that an ionic liquid could be a naturally occurring substance, considering the complexity of biochemistry. Two other possible sources, Davis said, are in sea sponges and the backs of poison dart frogs. The presence of ionic liquids would explain why the poisons of both are not washed off in their environments.
“The diversity of life and chemistry in living organisms is so vast that I can’t believe there are not other examples of this occurring,” Davis said. “We just didn’t know where to look.”
“Nature is an incredibly inventive sort of place, and there are all kinds of exotic chemicals and structures that exist. So in a sense I did not find it surprising when we saw that the combination of these two venoms produced an ionic liquid.”
Still unclear is how the ionic liquid protects the tawny crazy ant. Does it keep the fire ant’s poison from penetrating the tawny crazy ant’s exoskeleton? Does it detoxify the alkaloid-based venom?
The answer to those questions, and others, could help determine the practical implications of Davis’ discovery.
“With this discovery, I think there is a lot more that we will be doing in the area of natural-products chemistry as we continue to try to make new discoveries of ionic liquids in nature,” Davis said.
Archive Search
Latest University News
-

USA Launches New Hospitality and Tourism Concentrations
Global leadership and events marketing and management concentrations w...
April 17, 2024 -

Mortar Board Receives National Award, Initiates New Members
South's chapter of Mortar Board has received the Ruth Weimer Mount Cha...
April 17, 2024 -

USA Administrator Named as Faculty at National Institute
Dr. Mike Wilkinson has been chosen as a faculty member for 2024 New Pr...
April 16, 2024 -

South Earns National Designation for Cybersecurity Research
USA's School of Computing awarded a designation as a Center of Academi...
April 16, 2024