 |
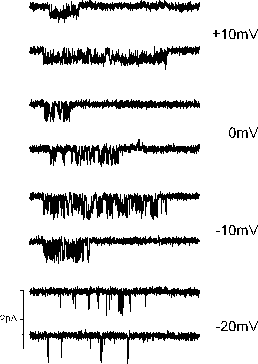
Examples of L-type Ca channel currents using 5 mM Ba ions, recorded at several test potentials.
Laboratory
of Cardiovascular Science
Gerontology Research Center
National Institute on Aging
National Institutes of Health
e-mail: JosephsonI@grc.nia.nih.gov
Introduction
This article is a
brief summary of some of the methods that are used to record single ion channel
activity in isolated heart cells. As
a representative example, the discussion will focus on techniques for recording
L-type Ca channel currents in isolated rat ventricular myocytes.
Using these methods we have found that single Ca channel activity can be
recorded using concentrations of Ba or Ca ions as low as 2 mM, and without the
need for any Ca channel agonists.
For a detailed and
comprehensive view of the subjects of recording and analyzing single ion
channels I recommend the following sources: Single Channel Recording by Neher
and Sakmann, the Axon Guide (available online at
www.axon.com),
and the thousands of research papers in the literature (a Medline
search for single or unitary ion channels or currents produced over 10,000
citations!).
Myocytes
For single channel recordings,
myocytes are usually isolated from rat or guinea pig hearts.
Of course other species may be used, depending on the nature and goals of
the study. However, before
selecting a species, one must keep in mind the relative abundance (or lack) of
the type of ion channel in question in myocytes of that species.
For example, if you wish to study Ito K channels you might select rat or
mouse, but not guinea pig ventricular myocytes.
We
have generally used rat myocytes for recording L-type Ca channels. Single rat
myocytes can be isolated by the following method. Male Sprague-Dawley rats (250
to 300g, 2 to 3 months old) were anesthetized with pentobarbital (80-100mg/Kg i.p.)
and their hearts were removed via a transverse incision over the diaphragm.
The hearts were washed in a nominally calcium-free modified Krebs
solution (in mM: 120 NaCl; 5.4 KCl; 1.6 MgSO4; 1 NaH2PO4;
20 NaHCO3; 5.6 glucose; 5 taurine; gassed with 95% O2, 5%
CO2) and then suspended and perfused via the aorta (constant
pressure, 100 cm H2O) on a heated (37°C) Langendorff apparatus.
The hearts were cleared of extracellular calcium by non-recirculating
retrograde perfusion of the same solution for 5 minutes then switched to a
recirculating solution of similar content with the addition of protease (0.02mg/mL,
type XIV, Sigma Chemical Co., St. Louis, MO, USA) and collagenase (1mg/mL; type
B, 220 to 230U/mg, Boehringer-Mannheim, Indianapolis, IN, USA, or
type 2, Worthington, Lakewood, NJ, USA), and, after 3 to 4 minutes in the enzymes 50µM
CaCl2 was added to the perfusate.
At the end of the first digestion, the ventricles were chopped into
several chunks and then placed into fresh Krebs solution containing 50 or 100µM
CaCl2 and collagenase (1mg/mL).
This second digestion was allowed to proceed in a shaker (60 to 70 rpm)
at 37 °C until a satisfactory yield was obtained (10 to 15 minutes).
The second digestion was quenched by filtering the supernatant for
centrifugation at 500G and three subsequent washes with a modified Tyrode
solution (in mM: 137 NaCl; 4.9 KCl; 15 Glucose; 1.2 MgSO4; 1.2 NaH2PO4;
20 HEPES; NaOH, pH 7.4) with successively increasing calcium concentrations
(250, 500 1000 µM). The cells were
allowed to settle from the supernatant for 10 minutes between washes. Cells were
stored at room temperature in a similar Tyrode solution containing 1 mM CaCl2.
The myocytes isolated in this manner were relaxed and rod-shaped, with clear
sarcomeric striations and smooth, clean membranes. (see the HELP
index for more detailed instructions on isolating cardiomyocytes from a variety
of species.)
Aliquots
of cells were placed in a 0.1 mL bath mounted on the stage of a conventional
inverted microscope. At least 10
minutes was allowed for the cells to attach to the coverslip on the bottom of
the bath. The cells were then
perfused with a high potassium depolarizing solution (HiK) at an approximate
rate of 3 mL per minute. The HiK
solution (in mM: 120 K-Aspartate; 25 KCl; 10 HEPES; 10 Glucose; 2 MgCl2;
1 CaCl2; 2 EGTA; 6 KOH, pH 7.2, 290 mOsm) was used to depolarize the
cells to near 0 mV so that Vm was equal to –Vpatch.
The free calcium concentration in the HiK solution was calculated to be
approximately 80 nM. To allow
stabilization in their new milieu, the cells were perfused with HiK for at least
20 minutes before unitary current measurements were conducted.
All experiments were performed at room temperature (22.5 to 23.5°C).
Borosilicate
pipettes made from Corning #7052 glass (1.5 OD, .86 ID, Model 5968, A-M
Systems, Inc., Carlsborg, WA, USA) were pulled in 3 or 4 heating cycles
using a horizontal Flaming-Brown pipette puller (model P-97, Sutter
Instrument Co., Novato, CA, USA) or a CO2 laser-based puller
(model P-2000, Sutter Instrument Co.) to
yield tips approximately 1µm in diameter.
The pipette tips were firepolished (model MF-83, Narishige
Instrument Lab., Tokyo, Japan) to produce 8 to 15 MOhm tip resistances when
filled with the pipette solutions, and were painted with a thick layer of
silicone elastomer (Sylgard, Dow Corning phone (800) 248-2481 or Catalog #184, Essex
Brownell, Fort Wayne, IN, USA) and polimerized under a heat gun to within 100µm of
the tip. Pipettes were filled with
a solution containing BaCl2 or CaCl2 of the desired
concentration, 10 mM CsCl and 5 mM 4-aminopyridine to block K currents, 10 mM
Hepes, with sucrose added to maintain normal osmolarity.
Pipettes were stored in a covered container and were back-filled with
pipette solution and used immediately. Seal
resistances of 50 to 300 Gigaohms were obtained by applying slight pressure with
the pipette tip on the membrane then applying gentle suction inside the pipette
using a Hamilton 1ml gas-tight glass
syringe connected to the top of the pipette with a meter of tygon tubing
(available from Fisher
Scientific). For
each seal, the pipette junction potential was offset to 0 mV with the pipette
positioned near the membrane before initiating a seal.
Formation of a stable seal was usually accomplished within a 20 to 30
seconds after the junction potential was nulled. Membrane and seal capacitances were corrected electronically.
The noise at a bandwidth of 5 KHz was measured and only seals quieter
than 250 fA RMS were used.
Current
amplification was accomplished with an Axopatch
200B patch clamp (Axon Instruments Co.) and recorded on a computer hard disk
using PClamp software
(v. 6 or v. 8, Axon Instruments Co.) via a
Digidata 1200A signal acquisition system.
Data was filtered at 2 KHz (-3dB, 4-pole Bessel) and digitized at 10KHz
sampling rate. Voltage-step protocols were applied at a rate of 0.5 Hz, from a HP
(holding potential) of –50 mV.
The step was varied from –50 to +30 mV (in 10 mV increments).
The protocol was repeated 100 - 200 times, or until channel rundown was
observed.
Analysis
Each file from a
series of repeated protocols was corrected for leakage and capacity currents by
subtraction of an average of episodes devoid of single channel activity during
the test voltage step (null sweeps). The
identification of single channel opening and closing transitions was
accomplished using Fetchan which is part of the Pclamp software package.
Events shorter than 0.2 msec were not included in the analysis.
The number of active Ca channels in a given patch was estimated by the
maximum number of overlapping currents (recorded at positive potentials).
The analysis of the probability of opening, open-time distributions and
their exponential fits, amplitude distributions and their gaussian fits, and
scatterplots of amplitude vs. duration were done using a modified version of
pSTAT supplied with the Pclamp software.

Examples of L-type Ca channel currents using 5
mM Ba ions, recorded at several test potentials.